Work should be shown for all calculations.
| 1. | Explain why the rate of a chemical reaction increases as the concentration of the reactants increases. |
|||||||||||||
| 2. | Consider two gases A and B in a container at room temperature. What effect will the following changes have on the reaction rate between these gases (increase, decrease, no effect)? a. The pressure is increased. b. The number of molecules of gas A is doubled c. The temperature is decreased |
|||||||||||||
| 3. | Which of the following reactions will have the fastest rate? The slowest? Explain | |||||||||||||
a. C12H26 (s) + 18.5 O2 (g) → 12 CO2 (g) + 13 H2O(g) b. S2O82-(aq) + 2 I–(aq) → 2 SO42-(aq) + I2 (s) c. Ba2+(aq) + SO42-(aq) → BaSO4 (s) |
||||||||||||||
| 4. | The series of steps by which an overall chemical reaction takes place is called the ________________. | |||||||||||||
| 5. | The slowest step in the series of steps in a chemical reaction is called the ____________________. | |||||||||||||
| 6. | Define activation energy. | |||||||||||||
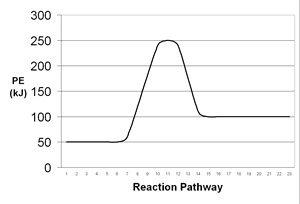
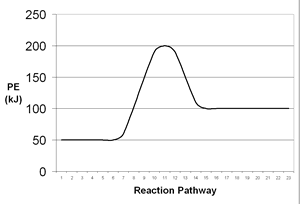
| 7. | Consider the following potential energy diagrams which represent two chemical reactions. On the basis of these diagrams, which reaction would you expect to occur at a faster rate? Why? | |||||||||||||
A |
B |
|||||||||||||
 |
 |
|||||||||||||
| 8. | Which will react faster, zinc with 3 M hydrochloric acid or zinc with 1 M hydrochloric acid? Why? | |||||||||||||
| 9. | White phosphorus reacts immediately and rapidly with oxygen when exposed to air. What can you say about the amount of activation energy required for this reaction? | |||||||||||||
| 10. | Hydrogen peroxide reacts with hydrogen ions and iodide ions according to the reaction mechanism shown below:
|
|||||||||||||
| 11. | At 20°C, a small strip of magnesium reacts with 3.0 M hydrochloric acid to produce 12 mL of hydrogen gas in 20 s.
|
|||||||||||||
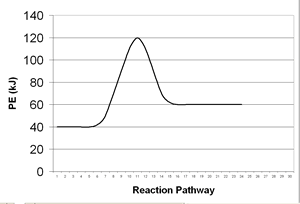
| 12. | Consider the following potential energy diagram:
|
 |
||||||||||||
| 13. | Sketch a potential energy curve for an reaction based on the information provided below.
Label the parts representing the activated complex, activation energy, and change in enthalpy, ΔH.
|
|||||||||||||