3.4 Electroplating
Our final example of electrolytic cells will examine a technique in which the electrodes actively participate in the reaction, instead of merely just carrying the electrical charge.
Electroplating is a technique in which a thin layer of a desired metal is used to coat (or "plate") another object. This process is often used to protect objects against corrosion or to improve their appearance.
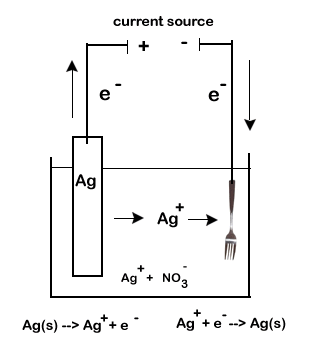
For our example we will exam the silver plating of flatware. In our example we will coat a fork made with an inexpensive metal with a thin layer of silver.
As with our other electrolytic cells we have three requirements:
- an electrolytic solution. Our electrolytic solution will need to contain ions of the plating metal. We will use AgNO3 which will give us our required Ag+ ions.
- a source of current (an electrochemical cell - a battery), and
- two electrodes. One of our electrodes will be the object to be coated (the fork), while the other must be the plating metal (a bar of silver).
Our half-reactions will be a bit different this time - both will involve the same reaction:
Here's the overall plan of action:
- We want solid silver to be deposited on the fork. Thus the fork electrode must be the cathode, or site of reduction. Positive silver ions from the electrolytic solution will be attracted to a negative electrode causing the deposition of solid silver.
- The Ag+ will come from the electrolytic solution. As they get used up, they will need to be replaced.
- The silver bar will be the source of new Ag+. The bar will act as the anode and undergo oxidation. As it disintegrate is produces new Ag+.
The final concern is to account for the flow of electrons. Remember, our electrolytic cell will be hooked up to a battery (a set of electrochemical cells) or other source of current.
- Electrons will leave the negative post of the battery (the anode).
- These electrons will travel through the external circuit to the cathode of the electrolytic cell (our fork, the cathode)
- The anode of the electrolytic cell produces more electrons as it undergoes oxidation. These electrons return to the current source.
Congratulations! You have now completed the reading material for this unit, and for the course. There is a final set of practice questions (Set 5) to complete, and a final assignment (Assignment 3) to do before writing the unit exam.